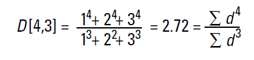Q1. Can these models be applied to a semi-solid formulation intermediate, e.g., in a melt spray congeal process where the API is distributed in a molten lipid and then congealed into spherical beads?
The models can be built based on the data collected on the resulting spherical beads. A heptane bases PSD method should work for a wet method, or a dry method could be used. Krypton based surface area could be used on the beads. Finally, ring shear flow instruments sell several sized sample cells to accommodate diversity in samples tested.
Q2. Bulk properties and API properties are directly related to the particle attributes notably morphology. Do you have an example in which you have used this information to tailor process parameters to obtain a desired morphology?
Per case study 3 discussion, as process DOEs or early RnD development investigations are conducted (including PAR and alt solvent ratios, stir speeds, etc) the API camera can be used to map the resulting affects on the yielded API. The data can then be used as road maps to target the quality attributes that may be of interest to support crystal engineering. Also please see my 2015 publication which touches base on this topic: Morrison et al, Org. Process Res. Dev., 2015, 19 (9), pp 1076–1081 (Correlation of Drug Substance Particle Size Distribution with Other Bulk Properties to Predict Critical Quality Attributes)
Q3. Morphology can be affected by impurities. Have you checked the purity of the API?
Yes, as programs progress though development, purity is monitored during RnD development and reported in the certificate of analysis for GMP lots.
Q4. What is VMD?
Volume mean diameter (AKA volume weighted mean, AKA D[4,3]) is the mean of a particle size distribution weighted by the volume. It is the mean diameter, which is directly obtained in particle size measurements, where the measured signal is proportional to the volume of the particles. It provided insight into the center of gravity for a distribution that is multi-modal.
Q5. Did you characterize the Bulk density of API and its impact on flowability of the powder?
Bulk and Tap density data was collected as well as the Carr index calculated. I have tried to model these parameters to PSD and SA; however, to date I have been unable to find a successful correlation between these parameters.
Q6. Any way to troubleshoot problems related to static?
On large scale batches (specifically milled lots) this can be a problem and attributes to the cohesiveness of a powder. Unfortunately I don’t have much experience with this phenomena outside of troubleshooting small powder aliquots with de-static devices.
Q7. For one case we observed that larger PSD (D90) of API can reduce the dissolution variation of tablets and increase the dissolution. Do you have any thoughts on that?
I would suspect that it might be related to cohesiveness of the API having smaller PSD values. My suggestion would be to correlate the PSD to flowability of the lots in question to see if there are differences that can be further correlated to the dissolution observations.
Q8.Slide 11: What was the drug load in the formulation in case study 1?
~30%
Q9. How applicable is the API camera to DP?
The principles of the methodology could be applied to dry blends for insight and understanding
Q10. The Morphologi G3 dispersion unit can cause breakage of acicular crystals due to the high air flow and impact of crystals on the unit. This can have a large effect on PSD and aspect ratio measurements. Did you investigate this, and would you recommend manual dispersion of crystals for analysis?
For the purposes of these investigations, we hand prepped our samples on microscope slides by dispersing the API in oil and using the microscope slide stage on the G3.
Q11. What was the drug loading for the tablet formulations in these case studies? Could the limitations of the API bulk properties be addressed using formulation and/or process fixes?
Study 1 = ~30%, Study 2 = ~20%, Study 3 = ~10%. For high drug load DP, sometimes it can be challenging to overcome the properties of the API via the formulation process. But yes, it is possible if you can formulate with appropriate glidants (however, you are limited by how much glidant can be used). As mentioned in case study 3, the models presented can act as a road map to target quality attributes based on the process experience; therefore, there are opportunities to overcome issues via process optimization for a program need.
Q12. What are some common techniques to improve flow problems?
1) grow larger particles
2) attempt to change morphology via crystal engineering
3) appropriate glidant in the DP blends
Q13. Can you comment on the difference and advantage of your mini-dissolution method for API and the more commonly used intrinsic dissolution method?
Both can be used and are equality valuable. Intrinsic dissolution requires manipulation of the API into a disk surface, therefore you can potentially loose insight that you might gain from allowing the free-flowing powder to distribute within the dissolution media which is often corelated to the cohesiveness of the powder itself.
Q14. Can you comment on ring shear flow measurement vs. simpler methods such as angle of repose or Hausner ratio?
I have not personally investigated the angle of repose method before, but I certainly understand that Ring Sear Flow instruments may not be commonly available, therefore I think it merits investigations to see if API Camera flow models can be built using this. I have attempted to model the bulk properties I spoke about with Bulk and Tap density data (and there for the Hausner ratio); however, to date I have been unable to find a successful correlation between these parameters via modeling.
Q15. The case studies showed the tripods are working collectively, so do we need to set API specification that includes PSD, surface area and powder flow?
My recommendation would be No. These tests should be measured as For Information Only (development tests). The models that can be generated via the API Camera can then be utilized to optimize a process so that teams can consistently target the quality attributes identified to support successful DP manufacturing.
Q16. Did you try using calculated particle surface area instead of BET surface area? Could that be an alternative?
Most PSD instruments can calculate a surface area; however, since the laser diffraction units are blind to morphology, the data is useless as applied to the API Camera models. Image based PSD units can attempt to do the same thing, but again, the data is based on 2D images and can provide not context on surface texture or porosity.
Q17. Can you please suggest the modeling software?
Machine learning methods, statistics, and data clustering were performed using Python 3.9.0 (Python Software Foundation), employing the statsmodels v0.12.2 library and scikit-learn 0.24.1 library.
Q18. How to work around for moisture sensitive APIs?
You can work around moisture sensitive APIs using lower moisture/less hygroscopic excipients, manufacturing in low relative humidity-controlled rooms, storage of intermediate products (e.g., preblends, final blend, bulk tablets) under dry conditions, and decreasing environmental moisture availability through packaging.
Q19. Were contact angle measurements or surface measurements done?
At the time of these investigation, a contact angle instrument was not available. We have since on boarded one and are actively attempting to evolve the API camera methodology by adding this technique as an added accessory.
Q20. While modifying bulk properties needed for DP, have you seen changes in dissolution rate of the API?
Yes, morphology differences can significantly affect dissolution when comparing samples of the same PSD but different shapes (again SA is then a better surrogate to predict dissolution). Also, as discussed in the presentation, PSD distribution shapes and their relationship to their resulting surface area will certainly affect dissolution.
Q21. Have you used this API camera model to predict flow ability of micro particle suspension? Any insights that you can share?
The models have been used to map and predict the flowability of the micro particles used to generate the suspensions, but not the suspensions themselves. We have recently used the API camera to trouble shoot differences in resulting API lots coming from 2 different jet mills to show that although both mills provided lots of the same PSD, the lots have very different cohesiveness (and wettability via our new contact angle instrument) as a function of the milled used. The data therefore explained why Mill A lots formed pastes when formulated into suspensions, while Mill B lots formed injectable suspensions.
Q22. To improve the bioavailability particle size reduction is preferable, or crystal modification like salt or cocrystal?
Both should be investigated. Typically, a free base/acid is investigated in early development and bioavailability challenges can be overcome as a function of particle size reduction. However, this can buy you only so much of an improvement that if you still do not hit the targeted exposure of interest, crystal modification should be investigated. At which point you can again apply particle size reeducation to the new salt phase should it be needed.
Q23. How do we calculate VMW?
Volume weighted mean is calculated by the instrumental software and is reported by its alternate name of D[4,3].
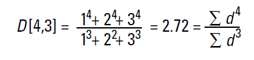
Q24. For the G3 Morphologi, if the API is too cohesive, can you disperse the primary particles to avoid measuring the morphology of agglomerate?
Samples can be prepared on microscope slides by dispersing the API in oil and characterized using the microscope slide stage on the G3. Having said that, true agglomerates represent a bulk property and should be accounted for to understand lot to lot differences.
Q25. Please discuss the model's ability to take into account the probability of polymorphic interconversion during milling, blending, granulation, and compression steps.
The models will not provide insight into the probability of polymorphic conversions, but the data can be correlated to the affect these conversions have on the bulk properties. Behind the scenes of the API camera, XRPD, DSC, TGA, etc are utilized to characterize the various API batches, and when polymorphic conversions occur, the resulting bulk property data points can often show up as an "orange in the basket of apples" when the curves are built on lots that are based on Form I and then a data point of Form II is modeled within the Form I curves.
Q26. Please discuss the model's ability to select among the following tableting methods: dry granulation, wet granulation, and direct compression.
The models will not necessarily drive the decision for selecting the tablet making process, but rather determine if the API properties are amicable to the various routes.
Q27. How would the model benefit from a better first-principles characterization of the API crystal structure?
It's possible that density functional theory could give insight to possible morphological possibilities of the crystal and potentially their surface related attributes, but at this time I believe this is a yet unexplored space worth looking into.